Difference between revisions of "ClinicalUseIssue FHIR Resource Proposal"
Riksmithies (talk | contribs) |
|||
| (3 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
| + | <div class="messagebox cleanup metadata"> | ||
| + | <div style="float: left;">[[Image:OpenHotTopic.GIF|35px| ]]</div> | ||
| + | <div style="background:#F0F0F0"> | ||
| + | This page documents an [[:category:Approved FHIR Resource Proposal|Approved]] [[:category:FHIR Resource Proposal|FHIR Resource Proposal]] | ||
| + | </div> | ||
| + | </div> | ||
| + | [[Category:FHIR Resource Proposal]] | ||
| + | [[Category:Approved FHIR Resource Proposal]] | ||
| + | |||
| + | <!-- For additional guidance on considerations for resource creation, refer to [[FHIR Resource Considerations]] --> | ||
| + | |||
=ClinicalUseIssue= | =ClinicalUseIssue= | ||
| Line 86: | Line 97: | ||
==Example Scenarios== | ==Example Scenarios== | ||
| − | Uses of these resources are in several categories | + | Uses of these resources are in several categories: |
Clinical trial systems that may register the discovery of a new side effect of a drug. | Clinical trial systems that may register the discovery of a new side effect of a drug. | ||
| − | Systems that directly exchange clinical use issues, such as when registering a newly invented drug with a regulator | + | Systems that directly exchange clinical use issues, such as when registering a newly invented drug with a regulator. The indications and unwanted effects are declared, and updates are made when necessary e.g. a new contra-indication. |
| − | Clinical Knowledge System vendors that collate prescribing support data (e.g. as published by regulators and manufacturers), and supply it to system | + | Clinical Knowledge System vendors that collate prescribing support data (e.g. as published by regulators and manufacturers), and supply it to system integrators (e.g. First Data Bank) |
Systems that use these information items in decision support processes. | Systems that use these information items in decision support processes. | ||
| Line 98: | Line 109: | ||
In fact data would originate with manufacturers (based on clinical trials), flow to regulators, then become available to clinical knowledge vendors, and them be integrated into EHR decision support systems. (In turn new side effects may be discovered in clinical care and recorded.) | In fact data would originate with manufacturers (based on clinical trials), flow to regulators, then become available to clinical knowledge vendors, and them be integrated into EHR decision support systems. (In turn new side effects may be discovered in clinical care and recorded.) | ||
| + | Specific use cases include: | ||
| − | + | Submission of products from drug companies and NCAs (National Competent Authorities - the national regulators) to regional regulators. | |
| − | |||
| − | |||
| − | + | This is already implemented in Europe (by EMA and EU-wide stakeholders) with an earlier non-HL7 format (XEVPRM/XEVMPD). | |
| − | + | That scenario is currently being re-implemented, using this resource, as part of the EU wide SPOR project. | |
==Resource Relationships== | ==Resource Relationships== | ||
| Line 112: | Line 122: | ||
Some notable resource references: | Some notable resource references: | ||
| − | + | ||
| + | Subject reference to MedicinalProductDefinition, Medication, Device/DeviceDefinition, and Activity-/PlanDefintition - the treatment that this is an indication for, for instance. | ||
===ClinicalUseIssue and MedicationKnowledge=== | ===ClinicalUseIssue and MedicationKnowledge=== | ||
| − | MedicationKnowledge resource is aimed at drug knowledge bases. | + | MedicationKnowledge resource is aimed at drug knowledge bases. These consist of many data points and it well refer to potentially severan ClinicalUseIssue instances. In general, where information about indications, contra-indications etc is needed, it is anticipated that this resource would be used. |
The boundaries between all these resource have been carefully thought out and have had much discussion in workgroups (BR&R, Pharmacy, CDS) and with FMG representatives. | The boundaries between all these resource have been carefully thought out and have had much discussion in workgroups (BR&R, Pharmacy, CDS) and with FMG representatives. | ||
Latest revision as of 21:07, 4 September 2019
Contents
- 1 ClinicalUseIssue
- 1.1 Owning work group name
- 1.2 Committee Approval Date:
- 1.3 Contributing or Reviewing Work Groups
- 1.4 FHIR Resource Development Project Insight ID
- 1.5 Scope of coverage
- 1.6 Resource appropriateness
- 1.7 Expected implementations
- 1.8 Content sources
- 1.9 Example Scenarios
- 1.10 Resource Relationships
- 1.11 Timelines
- 1.12 gForge Users
- 1.13 When Resource Proposal Is Complete
- 1.14 FMG Notes
ClinicalUseIssue
Draft resource in build:
Owning work group name
Committee Approval Date:
6th May 2019
Contributing or Reviewing Work Groups
- Pharmacy
- Orders and Observations
- Clinical Decision Support
FHIR Resource Development Project Insight ID
1367
Scope of coverage
ClinicalUseIssue covers the background information about potential use of treatments or therapies (drugs, devices, procedures, substance etc), and how they affect a patient, positively and in particular negatively.
The high level scope is:
Indications - in what circumstances it is good practice to give the treatment
Contraindications - in what circumstances it is NOT good practice to give the treatment
Undesirable effects - what possible negative consequences are known to be issue for this treatment ("side effects")
Interactions - what possible negative effects may be brought about by using this treatment (especially a drug), due to other previously employed drugs or treatments.
These are not related to any particular instance of use and also not any actual good or bad outcome, but are reference information, to guide the potential choice of treatments.
There is one resource for one set of facts about a single usage pattern. This does not cover multiple indications, or both indications and contraindications in one.
It is a polymorphic resource, that can be one of several distinct things - an indication and interaction are very different things. (Earlier iterations had 4 separate resources, but these were combined at the request of CDS WG).
Out of scope are actual events such as AdverseEvent (or Condition), and context specific collections of information such as MedicationKnowledge or MedicinalProductDefinition (both of which will use these resources).
These are also different to DetectedIssue, which represents a decision support finding being triggered. Detectedissue may well use these resources to indicate the source of information that was used in the activation process.
Resource appropriateness
The clinical concepts are well known, and well integrated into modern EHRs. Decision support systems are becoming mainstream, but may use proprietary formats to represent their source information. There is a clear need for this data in current generation systems, and support for it within FHIR seems highly appropriate.
This resource has been designed in consultation with CDS WG and Pharmacy WG, and in conjunction with the MedicationKnowledge resource.
Expected implementations
EMA and European drug manufacturers, who have a requirement to submit to EMA (and already do so in a proprietary format). They are required to move to IDMP, and this is a good opportunity to use a standards-based FHIR solution.
FDA for drug submission (currently using SPL, which is not likely to change in the near term, but have expressed an interest in FHIR).
FDA for Pharmaceutical Quality (HL7 PSS approved, based on this resource, June 2019),
Content sources
The core basis for the resource is the information in ISO 11615 Medicinal Products standard, which is in turn partly based on the existing implementations in the EU and US. A large amount of actual data exists in the EMA EU XEVMPD data base (and XEVPRM XML messages). Example FHIR data for several full product data sheets exists based on draft resources.
Also, information gained from early stage implementation of these resources at EMA (2018, 2019), and from many many received to EMA about the draft API specification from the European medicines regulatory network (https://www.ema.europa.eu/en/about-us/how-we-work/european-medicines-regulatory-network).
Also from FDA requirements (for PQ/CMC) and other workgroup review (BR&R, Pharmacy) and their comments.
Example Scenarios
Uses of these resources are in several categories:
Clinical trial systems that may register the discovery of a new side effect of a drug.
Systems that directly exchange clinical use issues, such as when registering a newly invented drug with a regulator. The indications and unwanted effects are declared, and updates are made when necessary e.g. a new contra-indication.
Clinical Knowledge System vendors that collate prescribing support data (e.g. as published by regulators and manufacturers), and supply it to system integrators (e.g. First Data Bank)
Systems that use these information items in decision support processes.
In fact data would originate with manufacturers (based on clinical trials), flow to regulators, then become available to clinical knowledge vendors, and them be integrated into EHR decision support systems. (In turn new side effects may be discovered in clinical care and recorded.)
Specific use cases include:
Submission of products from drug companies and NCAs (National Competent Authorities - the national regulators) to regional regulators.
This is already implemented in Europe (by EMA and EU-wide stakeholders) with an earlier non-HL7 format (XEVPRM/XEVMPD).
That scenario is currently being re-implemented, using this resource, as part of the EU wide SPOR project.
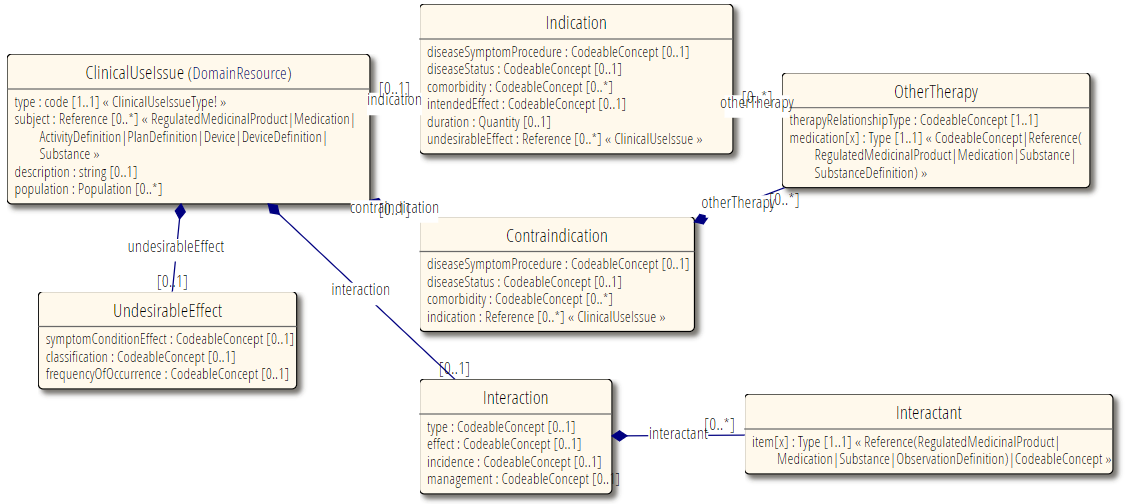
Resource Relationships
See diagram below.
Some notable resource references:
Subject reference to MedicinalProductDefinition, Medication, Device/DeviceDefinition, and Activity-/PlanDefintition - the treatment that this is an indication for, for instance.
ClinicalUseIssue and MedicationKnowledge
MedicationKnowledge resource is aimed at drug knowledge bases. These consist of many data points and it well refer to potentially severan ClinicalUseIssue instances. In general, where information about indications, contra-indications etc is needed, it is anticipated that this resource would be used.
The boundaries between all these resource have been carefully thought out and have had much discussion in workgroups (BR&R, Pharmacy, CDS) and with FMG representatives.
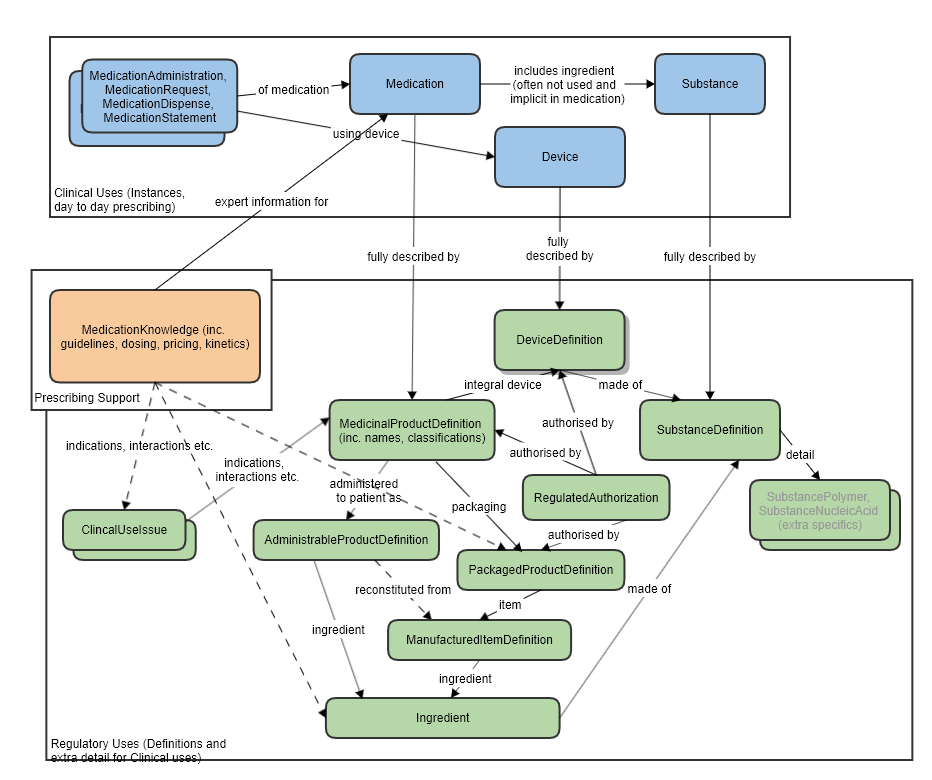
Also refer to the logical model which was used to clarify the resource relationships, at the request of FMG, in the preparation of this proposal (linked to the approved MedicationKnowledge proposal page): MedicationKnowledge_FHIR_Resource_Proposal
High level relationships of the main prescribing resources and the regulatory strata below:
Timelines
Draft content is modelled in the FHIR build (http://build.fhir.org/clinicaluseissue.html), with outline supporting documentation. Completion planned Q4 2019.
gForge Users
riksmithies
When Resource Proposal Is Complete
When you have completed your proposal, please send an email to FMGcontact@HL7.org